Introduction
The philosophical basis of informed consent lies in the concept of autonomy of individuals.(1,2) It is implicit that orthodontists treat patients with respect and act in their best interests. Orthodontic treatment is an amalgamation of various objective and subjective factors. While objective factors are derived from diagnosis and are definitive, subjective factors depend primarily on aesthetics and psychological considerations.(3) The Orthodontists view of the subjective issues may vary considerably from that of patient to parents. So to come to an agreement regarding various procedures, there should be an open dialogue between orthodontist and patient-parent party.(4) Seeking and obtaining this perspective in a clinical setting is possible through Informed Consent.
What is informed consent?
Informed consent is the process by which the treating health care provider discloses appropriate information to a competent patient so that the patient may make a voluntary choice to accept or refuse treatment.(5) It is the expression of the ethical principle of 'Respect for Persons' and 'Autonomy'. Here he/she has the opportunity to “choose what shall and shall not happen to them.”
Background
Notable historical cases of research abuse have influenced the development and regulation of informed consent processes. The discovery of unethical research and the resulting public outcry contributed to the institution of informed consent policies in research.6 The evolution of the research ethics guidelines is as follows:
- In 1900, Surgeon General George Miller Sternberg organized a medical commission headed by Dr. Walter Reed to investigate Yellow Fever in Cuba. No animal models were used and members of the board decided on human experimentation. This was the first example where consent in the form of contract was taken from the soldiers.
- 1932-1972: The Tuskegee Syphilis Study, sponsored by the U.S. Department of Health studied the effects of untreated syphilis in 400 African American men. Researchers withheld treatment even when penicillin became widely available and subjects were not informed about being part of an experiment. In 1979, the National Commission releases The Belmont Report, principles of ethical research on human subjects. The Report becomes a key document in human research ethics regulations in the U.S.
- 1939-45: German scientists conduct research on concentration camp prisoners. In 1947, The Nuremberg Code for research on human subjects was adopted.
- 1964: World Medical Association introduced Helsinki Declaration which was a set of ethical principles for research on human subjects. It has been revised several times, most recently in 2001.
- 1990: The US launches the Human Genome Project, a $20 billion effort to map and sequence the human genome which has its own ethical and social implications.
- 2010: The National Science Foundation (NSF) announces Responsible Conduct of Research (RCR) training requirements for funded investigators, students, and trainees. The National Institute of Health (NIH) expands and strengthens its RCR training requirements.
Elements of informed consent
Four elements
- Disclosure
- Comprehension
- Voluntariness
- Documentation
Disclosure
The following contents should be disclosed in the informed consent form:
- The nature and purpose of all viable treatment options and this includes implications of non-treatment.
- The proposed benefits and risks of treatment.
- Limitations of the proposed treatment and the likelihood of success.
- Important practical information that patients/parents need like an estimation of treatment time, the frequency of appointments, the need for additional appointments if breakages occur and the need for retention.
- The cost of the treatment (if applicable).
According to the American Association of Orthodontists, an informed consent for Orthodontic treatment should consist of the following details: results of treatment, length of treatment, presence of discomfort, chances of relapse, extractions; if needed, orthognathic surgery; if needed, occurrence of decalcification and dental caries, possibilities of root resorption; nerve damage, periodontal diseases; injury from orthodontic appliances, prospect of temperomandibular joint dysfunction, presence of impacted;ankylosed;unerupted teeth, occlusal adjustments; if any, non-ideal results, status of third molars, allergies, general health problems, consequences of use of tobacco products, use of temporary anchorage devices for treatment. It should also contain attached copies of acknowledgement, consent to undergo orthodontic treatment, authorization for release of patient information and consent to use of records.7
Comprehension
It is the process of enhancing and assessing potential participants' understanding of information which can be done in following ways:
- Using simple language as well as context specific (culturally?specific) analogies and metaphors.
- Providing information through accessible textual and non?textual means. For example: audio-visual aids and information leaflets.
- Developing the consent form/information in the language that it will be easily communicated.
- Pilot testing informed consent procedures by asking questions or conducting short quizzes.
- For individuals under sixteen years of age, parents or guardians can sign the informed consent.
- For individuals who are incompetent in nature, the decision can be taken by parents or guardians.
Voluntariness
It should be clearly established that consent is provided without any external influence or pressures.
Documentation
It is better to have a record that the process has been implemented for reasons of legal protection and to keep track of progress of treatment.
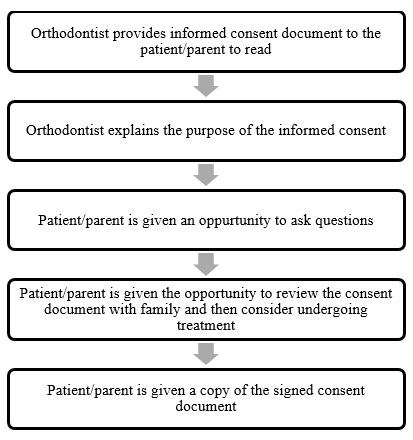
Informed consent process
Process can vary but should generally include
Problems of informed consent
- In some contexts, practitioners are expected to (and themselves may expect to) make decisions for patients - Paternalistic approach. Thus, lay persons and practitioners may find informed consent an alien concept.
- In many situations, written consent may be inappropriate when patients/parents are illiterate.(8)
Conclusion
The informed consent promotes the rights of a patient as autonomous beings to ensure that they are treated with justice, beneficence, and respect. Neglecting its importance can lead to unethical behaviour and the loss of patient's rights. Nowadays patients no longer want to be treated as passive recipients of medical care. Medical litigation and demands for medical accountability is the trend of the day. Ultimately, orthodontic treatment aims to provide the patient with optimum dento-facial aesthetics and a functional, stable occlusion.(9) If these principal factors are to be achieved, shared responsibility between the orthodontist and the patient must be sought and valid consent obtained prior to active treatment. Consent issues can be complex particularly with respect to the treatment of children and are constantly changing. It is imperative that clinicians maintain an up-to-date knowledge of the legal aspects of consent so that optimum clinical care can be delivered.
References
- Chate RAC. An audit of the level of knowledge and understanding of informed consent amongst consultant orthodontists in England, Wales and Northern Ireland. Br Dent J. 2008;205(12):665-73.
- Jayne E. Orthodontic Clinical Trials III: Reporting of ethical issues associated with clinical trials published in three orthodontic journals between 1989 and 1998. J Orthod. 2005;32(1):115-21.
- James L, William R. Communication in orthodontic treatment planning: Bioethical and informed consent issues. Angle Orthod. 1995;65(4):253-62.
- Sánchez S, Salazar G, Tijero M, Diaz S. Informed consent procedures: Responsibilities of researchers in developing countries. Bioethics. 2001;15(3):398-412.
- Appelbaum PS. Assessment of patient's competence to consent to treatment. New Eng J Med. 2007; 357: 1834-1840
- David B. Research ethics timeline (1932-Present). NIEHS; 2015.
- Fact sheet: Informed consent for the orthodontic patient: Risks and limitations of orthodontic treatment. USA: American Association of Orthodontists; 2005.
- Wendler D, Grady C. What should research participants understand to understand they are participating in research? Bioethics. 2008;22(4):203-208.
- Sander B, Alfred G, Bram T. Value of informed consent in surgical orthodontics. J Oral Maxillofac Surg. 2009;67(4):1021-25.